These Regulatory Affairs Team OKR templates are meant to help teams move from ideas and projects to measurable business outcomes. Use them as a starting point, then tailor the metrics and initiatives to the reality of your company.
Use Regulatory Affairs Team OKRs to define what success looks like this quarter, then track them weekly so the team can quickly spot blockers, learn, and adjust execution.
This page shows the top 3 of 3 templates for regulatory affairs team, with internal links to related categories and guidance for adapting the examples to your team.
Last template update in this category: 2024-08-22What this category is for
- Teams that need a clearer operating rhythm for regulatory affairs team work.
- Managers who want examples they can adapt into outcome-focused quarterly plans.
- Leaders comparing adjacent categories before choosing the best OKR direction.
Best outcomes to track
- Regulatory Affairs Team priorities tied to measurable business outcomes.
- Weekly check-ins that surface blockers before they become delivery issues.
- Better alignment between initiatives and the metrics that matter.
Related categories
Use these linked categories to explore adjacent planning areas and strengthen the internal topic cluster around regulatory affairs team.
Regulatory Affairs Team OKR examples and templates
Start with these top 3 examples from 3 total templates in this category, then adapt the metrics and initiatives to fit your team's constraints and operating cadence.
OKRs to develop innovative pharmaceutical products
ObjectiveDevelop innovative pharmaceutical products
KRDevelop and test 2 prototypes with satisfactory patient outcomes
Conduct patient outcome tests on prototypes
Identify key components for prototype based on patient needs
Build and refine two prototype designs
KRConduct research to identify 3 potential active pharmaceutical ingredients
Identify key drugs suitable for pharmaceutical component research
Conduct thorough research on potential active ingredients
Document findings for each potential ingredient
KRSecure regulatory approval for 1 new pharmaceutical product
Respond promptly to regulatory feedback and additional information requests
Conduct rigorous clinical trials for safety and efficacy
Develop and submit detailed dossier to regulatory authority
OKRs to secure FDA approval for the capital equipment medical device
ObjectiveSecure FDA approval for the capital equipment medical device
KRComplete and submit comprehensive FDA application by week 4
Thoroughly fill out the FDA application form accurately and comprehensively
Gather all necessary documentation and information for FDA application
Submit the completed FDA application before the deadline in week 4
KRObtain confirmation of FDA approval by end of the quarter
Regularly follow-up with FDA regarding application status
Prepare and submit all necessary documentation for FDA approval
Ensure all response deadlines to FDA inquiries are met
KRPass all necessary FDA inspections successfully with zero citations
Implement strict internal quality control measures
Regularly inspect and document all processes for compliance
Review and comprehend all FDA guidelines and expectations
OKRs to secure FDA approval for our new pharmaceutical product
ObjectiveSecure FDA approval for our new pharmaceutical product
KRResolve all FDA queries or issues regarding the application within six weeks
Research and compile thorough responses to each issue
Submit all responses and corrections to FDA within six weeks
Identify all FDA queries or issues on the application
KRSubmit a complete and compliant application to FDA within the first month
Review FDA guidelines to ensure application compliance
Submit the completed application to the FDA
Gather all necessary documents and data for application
KRSuccessfully pass the FDA's inspection and audit of our production facilities
Ensure all documentation and records are accurate, updated, and easily accessible
Provide thorough training to staff on FDA regulations and requirements
Maintain the facility's cleanliness and safety according to FDA standards
How to use Regulatory Affairs Team OKRs well
Strong OKRs keep the team focused on measurable outcomes instead of a long task list. That means picking a clear objective, limiting the number of competing priorities, and reviewing progress every week.
Use Regulatory Affairs Team OKRs to define what success looks like this quarter, then track them weekly so the team can quickly spot blockers, learn, and adjust execution.
Choosing software to run these OKRs?
Many teams looking for regulatory affairs team OKR examples are also comparing tools to roll them out. If you want to move from examples to execution, review our OKR software comparison guide to compare the best OKR software before you commit to a platform.
Related OKR template categories
If you are building a broader plan, these related categories can help you connect regulatory affairs team work to adjacent company priorities.
- leadership OKR templates
- strategic planning OKR templates
- operations OKR templates
- operations team OKR templates
- sales OKR templates
- sales team OKR templates
More OKR templates to explore
OKRs to strengthen quality control for product consistency
OKRs to foster continuous improvement culture in production team
OKRs to develop a robust vendor management framework
OKRs to implement a centralized sales data repository and reporting system
OKRs to drive company transformation through HR technology and leadership model redesign
OKRs to assemble a skilled and efficient analytics team
Not seeing what you need?
Use Tability AI to generate OKRs based on a prompt
Tability allows you to describe your goals in a prompt, and generate a fully editable OKR template in seconds.
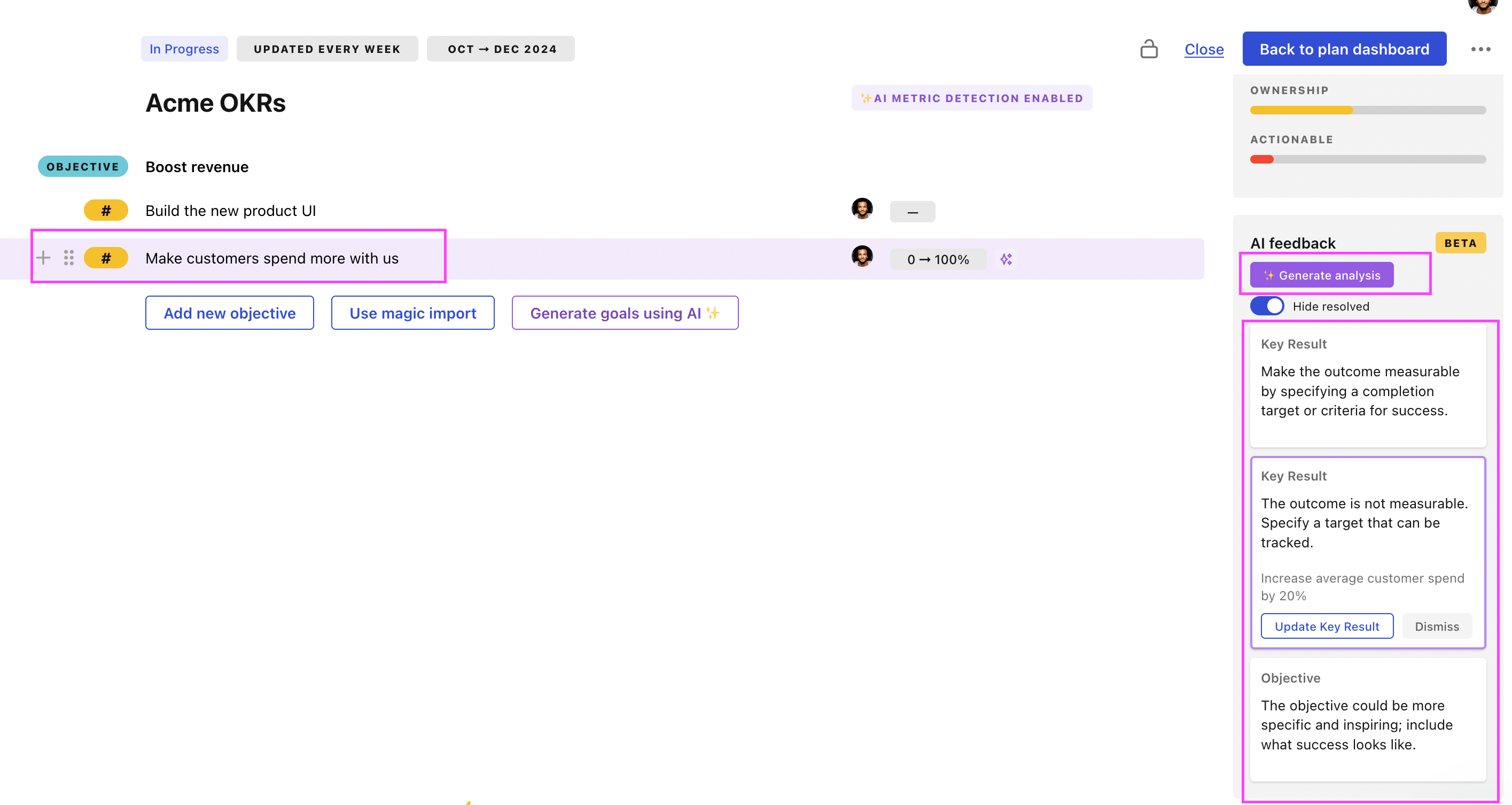
Use Tability feedback to improve existing OKRs
You can also use Tability's AI feedback to improve your OKRs if you already have existing goals. Just import them to the platform and click on the Generate analysis button.
Tability will scan your OKRs and offer different suggestions to improve them. This can range from a small rewrite of a statement to make it clearer to a complete rewrite of the entire OKR.